Lung Single-Cell Signaling Interaction Map Reveals Basophil Role in Macrophage Imprinting
1/17/2019
Merav Cohen, Amir Giladi, Anna-Dorothea Gorki, Dikla Gelbard Solodkin , Mor Zada, Anastasiya Hladik, AndrasMiklosi, Tomer-Meir Salame, Keren Bahar Halpern, Eyal David, Shalev Itzkovitz, Tibor Harkany, SylviaKnapp, IdoAmit
In this featured paper, Cohen et al. demonstrate how lung-resident basophils regulate alveolar macrophage maturation and immunomodulation functions.
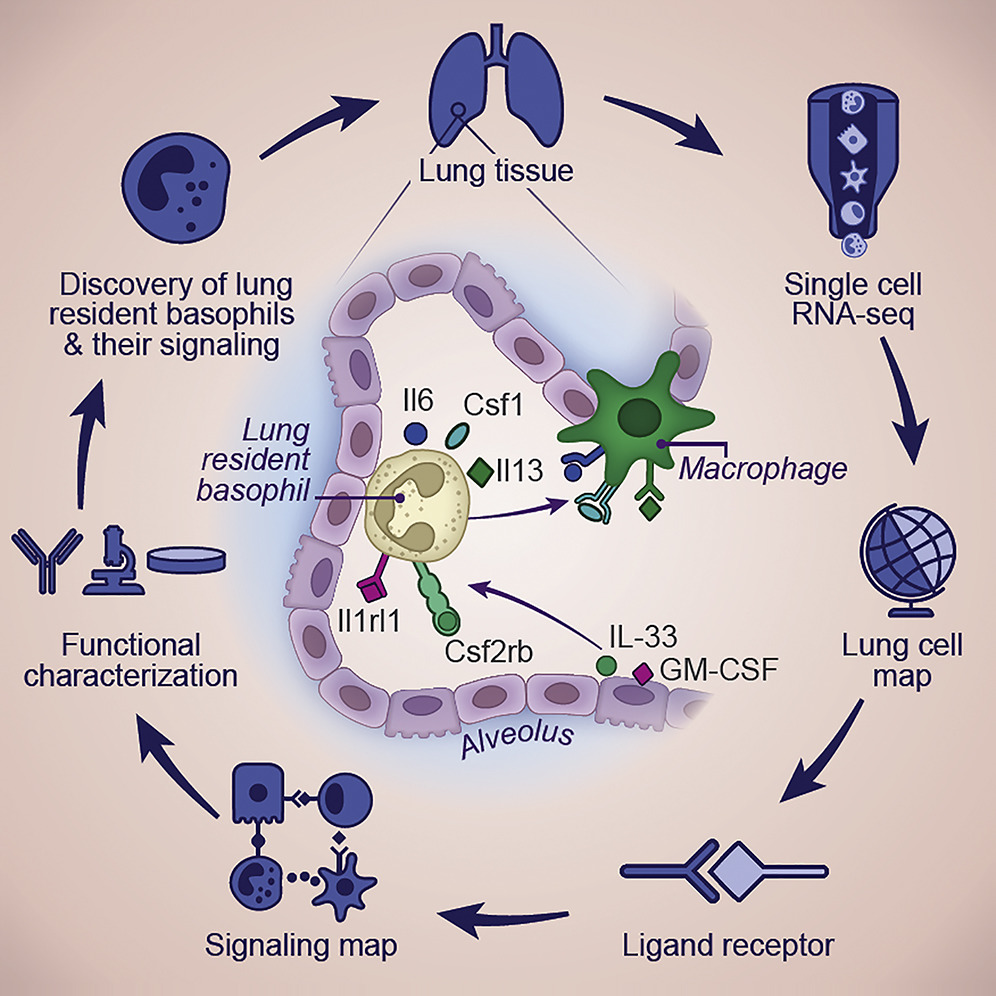
Lung development and function arises from the interactions between diverse cell types and lineages. Using single-cell RNA sequencing (RNA-seq), we characterize the cellular composition of the lung during development and identify vast dynamics in cell composition and their molecular characteristics. Analyzing 818 ligand-receptor interaction pairs within and between cell lineages, we identify broadly interacting cells, including AT2, innate lymphocytes (ILCs), and basophils. Using interleukin (IL)-33 receptor knockout mice and in vitro experiments, we show that basophils establish a lung-specific function imprinted by IL-33 and granulocyte-macrophage colony-stimulating factor (GM-CSF), characterized by unique signaling of cytokines and growth factors important for stromal, epithelial, and myeloid cell fates. Antibody-depletion strategies, diphtheria toxin-mediated selective depletion of basophils, and co-culture studies show that lung resident basophils are important regulators of alveolar macrophage development and function. Together, our study demonstrates how whole-tissue signaling interaction map on the single-cell level can broaden our understanding of cellular networks in health and disease.
Read more in: Cohen et al., 2018, Cell 175.4. November 2018.
 Immune & Epithelial Interactions
Immune & Epithelial Interactions Allergy & Asthma
Allergy & Asthma