Colonoscopy-based colorectal cancer modeling in mice
1/4/2018
Roper, Jatin et al. (2017). Colonoscopy-based colorectal cancer modeling in mice with CRISPR–Cas9 genome editing and organoid transplantation. Nature Protocols 13, 217–234, doi:10.1038/nprot.2017.136.
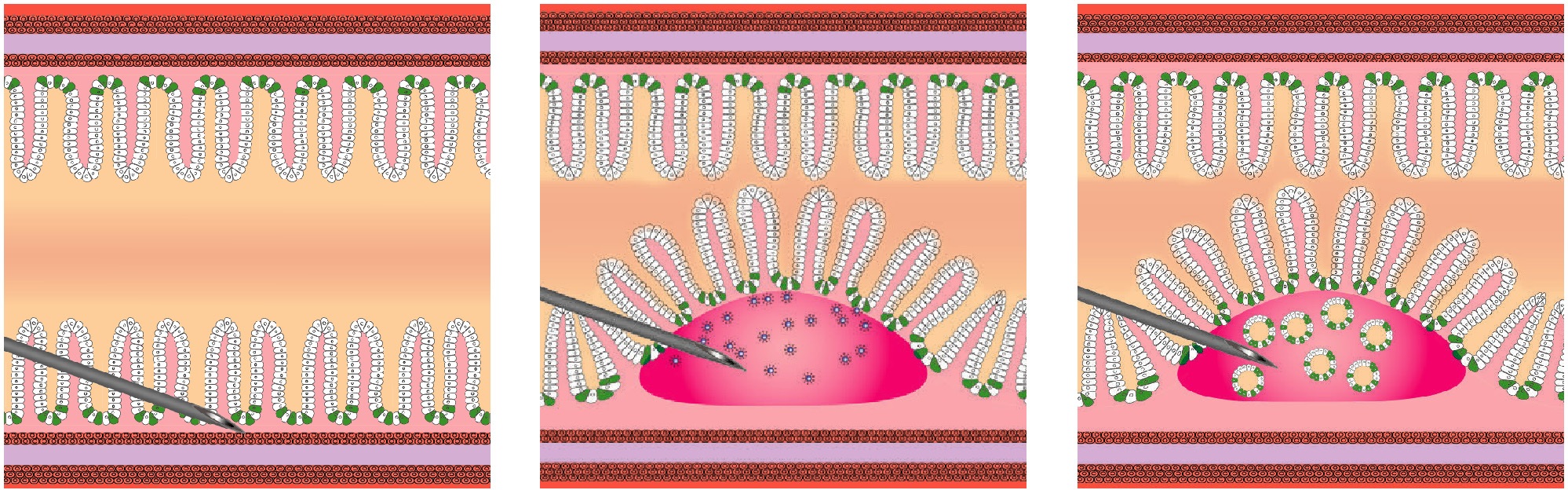
Most genetically engineered mouse models (GEMMs) of colorectal cancer are limited by tumor formation in the small intestine, a high tumor burden that limits metastasis, and the need to generate and cross mutant mice. Cell line or organoid transplantation models generally produce tumors in ectopic locations—such as the subcutaneous space, kidney capsule, or cecal wall—that do not reflect the native stromal environment of the colon mucosa. Here, Roper et al. describes detailed protocols to rapidly and efficiently induce site-directed tumors in the distal colon of mice that are based on colonoscopy-guided mucosal injection.
The use of colonoscopy-guided mucosal injection for CRC modeling was first reported by Zigmond et al. in 2011, who orthotopically transplanted mouse CRC cell lines with a 30-gauge injection needle and showed that the cells successfully formed tumors in the colon32. Since then, another group has applied this approach to model liver metastasis from orthotopically engrafted human CRC cell lines33.
Roper’s techniques can be adapted to deliver viral vectors carrying Cre recombinase, CR ISPR –Cas9 components, CR ISPR -engineered mouse tumor organoids, or human cancer organoids to mice to model the adenoma–carcinoma–metastasis sequence of tumor progression. The colonoscopy injection procedure takes ~15 min, including preparation. With Roper’s advice, anyone with reasonable hand–eye coordination can become proficient with mouse colonoscopy and mucosal injection with a few hours of practice. These approaches are ideal for a wide range of applications, including assessment of gene function in tumorigenesis, examination of tumor–stroma interactions, studies of cancer metastasis, and translational research with patient-derived cancers.
 Cancer
Cancer